How to Winterize a Commercial Swimming Pool
When summer ends, outdoor pools face an inevitable change in water chemistry. This change is driven by water temperature’s negative impact on water balance, as measured using the Langelier Saturation Index (LSI) or Ryznar Stability Index (RSI). This article covers how to winterize a commercial pool to prevent problems like etching, calcite crystallization, winter dust, and other problems.
Cold, hungry water tends to etch plaster
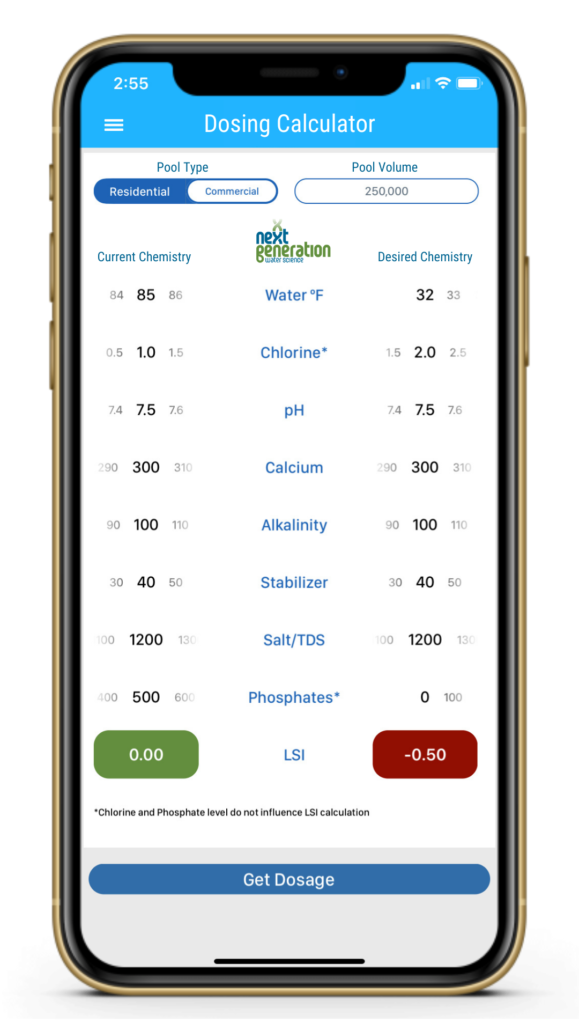
The water was perfectly balanced at 85ºF. Just lowering the temperature can make a huge impact on water balance.
Regardless if you use the LSI or RSI, colder water lowers each index, meaning water is more aggressive when it’s cold. We here at NextGeneration prefer using the LSI, which is why our free mobile app is called “LSI Calculator”. We can see from the following screenshot that just lowering the water temperature makes a huge difference in water balance, from 85ºF in the summer down to 32ºF in the winter. In the example shown, it’s an outdoor commercial pool in a region of the country that virtually guarantees freezing weather.
When the LSI is below -0.30, it means the water is starving for more calcium carbonate (CaCO3), and is looking for it anywhere it can be found. In pools with cement-based finishes like plaster, quartz or pebble, calcium is available in the surface. This is why most etching occurs in the winter, because the water is hungrier when it’s cold. This makes it even more important to winterize a commercial swimming pool correctly, with the LSI in mind.
When water etches plaster, the high pH of the calcium hydroxide taken from the surface (12.6 pH) spikes the pH in the pool. This is why it is not uncommon at all for pools to have an un-measurably high pH in the spring when re-opening. Naturally, pH can only rise up to about 8.2 thanks to Henry’s Law of physics. So if the pH far exceeds that, you know the pool was etched at some point during the winter.
Calcium is more soluble in cold water
Most substances dissolve easier in hotter water. Think of sugar or other additives when cooking. If you want to dissolve enough sugar to make a dessert, you probably have to heat the water to dissolve it, because room temperature water (or colder) will just have a pile of sugar staring back at you.
It’s the opposite with calcium.
Calcium is more soluble in cold water, which is why the LSI actually goes down as the temperature drops. This tells us the water is hungrier for more calcium, but it also tells us that calcium carbonate itself is more likely to be dissolved. Since plaster surfaces have an outer layer of calcium carbonate, etching goes after it. If the water dissolves through the carbonate layer, it has access to the much-more-soluble calcium hydroxide (Ca(OH)2).
How to winterize a commercial swimming pool
 There are two sides to water chemistry, and they both apply to winterizing too. There’s water balance (LSI, calcium saturation, etc.), and sanitization (keeping the water clean and clear). Our startup process covers both.
There are two sides to water chemistry, and they both apply to winterizing too. There’s water balance (LSI, calcium saturation, etc.), and sanitization (keeping the water clean and clear). Our startup process covers both.
1. Water balance
When you winterize a commercial swimming pool, the name of the game is keeping the LSI balanced throughout the winter. As we know, the colder the water, the more calcium it will crave. So in order to overcome this, simply use our free calculator app and input your chemistry on the left side, then adjust the desired chemistry on the right side so you are able to balance the water at the coldest temperature your pool could experience.
If you have the luxury of visiting the pool multiple times going into the winter, ease your chemistry up as the temperature drops rather than dumping in a huge amount of calcium in September when the water is only starting to cool down. But even if you do that, you run the risk of over-saturating the water with calcium carbonate, which leads to calcium precipitation, aka scale.
See the calculator screen below, where we lowered the temperature in preparation for a freezing winter. We then adjusted our calcium level and let the pH naturally rise up to its ceiling of about 8.2.
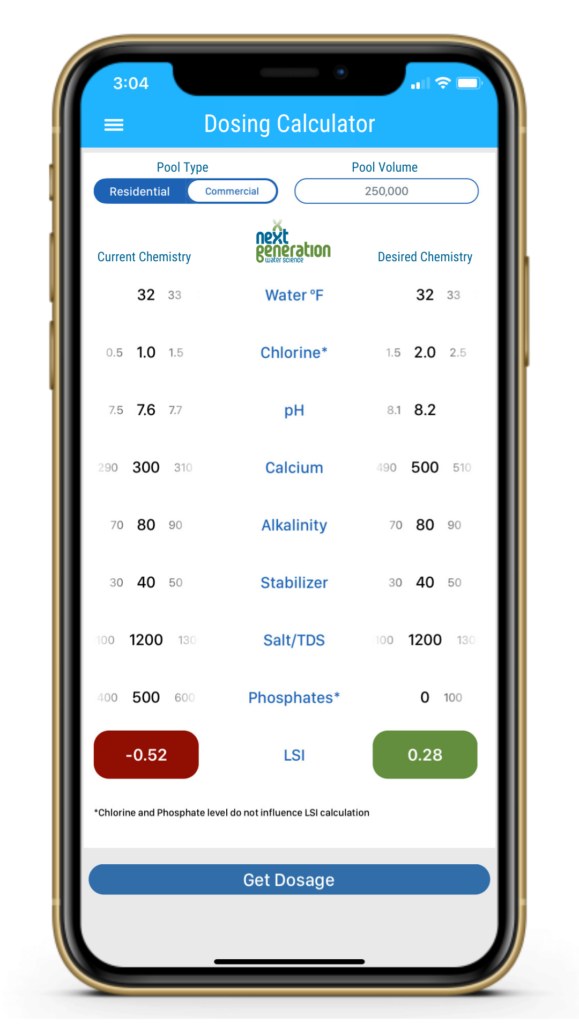
At 32ºF, the water was aggressive, despite being balanced during the summer. So we had to compensate for the cold water by substantially raising the calcium hardness level, and allowing the pH to naturally rise (thanks to physics) to its ceiling of about 8.2.
Does 500 ppm of calcium seem like a lot to you? To most people it does, until you factor in dilution. Unless your pool has a solid safety cover that allows no rain or snow into your pool, you can count on dilution to reduce your calcium hardness quite a bit. And depending on where you live, you could be contending with acid rain too. Rain and snow have zero calcium hardness, which makes precipitation extremely low and aggressive on the LSI.
If your pool is guaranteed to freeze, like outdoor pools in the American Midwest or Northeast, 500-600 ppm calcium hardness is going to be great for you. We say a minimum of 400 ppm if you have a solid safety cover. The right level depends on your circumstance, and we always say if you’re going to err, err high. Calcium is your best friend in the winter, so be generous with it. In residential pools calcium is needed too, but the volumes of water tend to be much less than commercial pools. The amount of calcium loss is proportional, but much less than, say, a 200,000 gallon pool. This is why we are focused on how to winterize a commercial swimming pool, and protect the surface from etching.
2. Sanitization
Fortunately, most living contaminants–like algae–have a difficult time reproducing in cold water. Usually, pools turn green from organics that get into the pool, like leaves and grass clippings. If the pool does turn green from algae, it’s usually the result of tannins from leaves and perhaps an algae bloom in the spring when the water is warming up once again.
In other words, you don’t have to chlorinate a pool all winter long. You just need to chlorinate properly at closing, and prepare the pool to minimize its risk of algae or other problems coming back. Many pools use algaecides, but we believe that’s unnecessary, since free chlorine itself is the best algaecide on the market. Instead, we opt to focus on removing phosphates and non-living organics. Neither phosphate remover nor enzymes are sanitizers, and neither of them will kill algae. They handle specific things that assist chlorine in other tasks so chlorine can keep the water clean and clear. Here’s our chemical recipe for winterizing, from a sanitization standpoint:
- Chlorinate normally (super-chlorination is not necessary unless your combined chlorine is above 0.2 ppm)
- Remove phosphates with CPR (about 8 oz. per 10,000 gallons) one week prior to closing day.
- Vacuum the precipitate and clean out filters after this treatment has had time to clear.
- Purge with AAD enzyme if the pool has not been using enzymes already during the season. If the pool is already on enzymes, use half a purge dose (16 oz. per 10,000 gallons) on closing day.
Step-by-step winterization procedure
- Use the free “LSI Calculator” app to balance the water at the coldest future winter temperature.
- If your pool will freeze, you will need a bare minimum of 400 ppm calcium hardness (if you have a solid safety cover), but we recommend more like 500-600 ppm calcium if you have a mesh safety cover or no cover at all. This extra calcium helps buffer against dilution.
- Pre-dissolve the prescribed amount of calcium chloride in buckets of water with a few ounces of MSI to chelate the calcium before adding it into the pool.
- Add up to 8 oz. per 10,000 gallons of CPR concentrated phosphate remover at least a day or two prior to closing.
- The pool will need to be vacuumed and the filters will need to be cleaned to remove the fallout dust.
- Purge the pool with 1 quart (32 fl.oz.) per 10,000 gallons of AAD enzymes.
- Chlorinate normally and proceed with other winterization tasks as normal. Maybe that includes lowering the water level, blowing out plumbing lines, covering the pool, etc. But avoid using antifreeze if possible, because AAD will conflict with them, and it can cause excessive foaming in the spring.
Conclusion
Focus on water balance as the temperature drops. To compensate for it, pre-dissolve and chelate calcium chloride with MSI, and get the calcium hardness level up to a high enough level to sustain the cold months. Account for dilution. If you know your pool will get snow and rain during the winter, more calcium will help you withstand the winter better than less. The LSI Calculator app will give you all the dosing information you need to winterize a commercial swimming pool successfully. If you have further questions, feel free to reach out. Thanks.


