Chlorine demand is a common concern for commercial pools. Is your swimming pool consuming more chlorine than you would like, or more than it should be? There are many reasons why this can happen, so in this article we’ll explain a few of them, and offer a remedy for each.
What reduces chlorine?
Chlorine has two primary functions in water chemistry: sanitzation/disinfection, and oxidation.

Reduction by Oxidation
The term “reduce”, or “reduction” of chlorine is the antonym to “oxidize”. The process of oxidation is the exact opposite of the process of reduction, which is why ORP (oxidation-reduction potential) is so commonly misunderstood. Oxidation is the loss of electrons by an oxidant and given to an oxidizer. Reduction is the gain of electrons by the oxidizer (chlorine or perhaps ozone or a hydroxyl radical). Because electrons (e-) are negatively charged, they reduce the valence of the oxidizer.
Chlorine in its powerful killing and oxidizer form, Hypochlorous Acid (HOCl) is reduced by electrons into weak chlorides (Cl-) that can no longer oxidize (steal more electrons). Therefore, that chlorine has been reduced, or used up. It’s not free chlorine anymore.
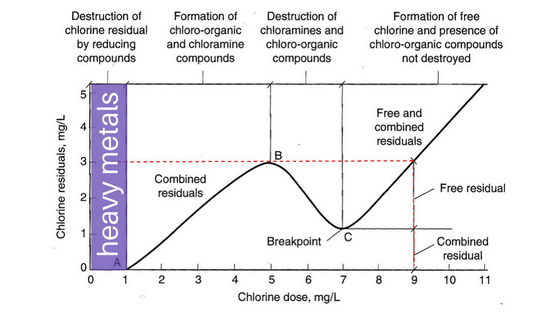
Metals are the first things to be oxidized when chlorine is added to water.
Compared to its strength as a sanitizer, chlorine is a relatively weak and inefficient oxidizer. Depending on what the oxidant is, its complexity determines how much chlorine will be consumed. Iron is easy to oxidize and occurs quickly. Sunscreen, on the other hand, is quite complex, and takes more chlorine to remove. And that’s nothing compared to a nitrogen compound like urea, which requires chlorine combining with ammonia and creating all sorts of disinfection byproducts (DBPs) like chloramines. These byproducts are measured as combined chlorine. See the breakpoint chlorination curve above.
Related: Water Chemistry Lesson: Combined Chlorine
Oxidation is chlorine’s method of eliminating non-living contaminants from water, like organic waste (bather waste, oils, sunscreen, cosmetics, lotions, etc.) and nitrogen compounds (ammonia, urea, etc.).
Reduction Consumption by Sanitization
Unlike oxidation, chlorine and other sanitizers do not steal electrons in the process of sanitization/disinfection, because the target contaminant is a living thing, not an oxidant. But like oxidation, chlorine also gets consumed in the killing process, it’s just not technically “reduced” because of electrons.
Sanitization is chlorine’s way of killing all living contaminants, and chlorine is really good at it. Killing germs and algae is what chlorine was made for. It is incredibly efficient at killing, whereas it is not so efficient at oxidizing complex non-living organics and nitrogen compounds.
Chlorine Demand
Let’s break down these demands into the two categories of sanitization and oxidation.
Sanitizer demand
As long as chlorine’s kill rate is greater than the growth (reproduction rate) of the contaminant, the water will not be at risk of outbreak.
Living organisms that reproduce will consume chlorine. Naturally, these will be more prevalent in warmer water, because things like algae grow better in warm water. Cold water is not conducive to algae or bacterial growth…or at least, not nearly as fast. The name of the sanitization game is making sure chlorine’s killing speed is faster than the contaminant’s reproduction rate.
Algae are a huge part of chlorine demand in outdoor pools. Algae not only consume chlorine, they also consume carbon dioxide, which raises the pool’s pH, causing increased acid demand too.
Algae growth is almost always a consequence of overwhelmed chlorine, either by overstabilization, or by an abundance of nutrients like phosphates.
Germs and parasites are far more of a concern for the health and safety of a swimming pool, but fortunately, there are very few of them in the water, relatively speaking. Most germs and viruses are killed very quickly by chlorine (again, provided the pool is not over stabilized with too much CYA). Parasites like Cryptosporidium, however, are notoriously hard to kill, even with very high levels of chlorine. The contact time (CT) required to kill parasites is almost not even comparable to simple germs and other bacteria. We could go on and on about recreational water illnesses (RWIs), but the CDC has already published the best RWI-prevention practices, and we are focusing on the consumption of chlorine for this article.
How to minimize sanitizer demand
To reduce the amount of chlorine consumed on germs, there are basically two ways you can attack the issue. First, and perhaps most obviously, is to optimize chlorine efficiency and strength. One way is to supplement chlorine with a secondary disinfection system like UV, Ozone or AOP. This will take some of the burden off chlorine by killing just about anything that passes through the pump room. But therein lies the weakness of secondary systems: they are point-of-contact only. They do not offer a residual that can circulate throughout the pool itself–a fact that reinforces the need for a primary residual sanitizer like chlorine.
Not all secondary disinfection systems are ideal for any pool. UV, for instance, is not ideal for an outdoor pool because of a particular organic waste that is common in the water: sunscreen. Sunscreen blocks UV light, and therefore limits the efficacy of UV on outdoor pools. They still help, but they are not nearly as beneficial as something like Ozone or AOP. Indoor pools, however, can benefit greatly from UV, though UV itself will gradually break down chlorine just like direct sunlight would.
Another option to supplement chlorine is with enzymes, though enzymes are not sanitizers. Enzymes handle the non-living organic oxidant demand–which happens to be the single-biggest category of contaminants in a pool–so there is more free chlorine available to kill germs. More on enzymes in a moment.
Optimizing chlorine isn’t just about strengthening it, it’s also about minimizing things that hold it back. Namely, cyanuric acid. CYA is beneficial for sunlight protection, but too much CYA dramatically weakens chlorine (%HOCl) and slows it down. Slow, weak chlorine means higher contact times (CT) when killing living pathogens.
 The second way to minimize sanitizer demand is to limit the growth and reproduction rate of algae. At any given time, an outdoor pool will be faced with algae spores trying to produce a colony. There is almost certainly more algae than there are germs, so we want to slow their growth and reproduction rates so chlorine can stay ahead. This can be accomplished by using Concentrated Phosphate Remover (CPR) as a proactive measure.
The second way to minimize sanitizer demand is to limit the growth and reproduction rate of algae. At any given time, an outdoor pool will be faced with algae spores trying to produce a colony. There is almost certainly more algae than there are germs, so we want to slow their growth and reproduction rates so chlorine can stay ahead. This can be accomplished by using Concentrated Phosphate Remover (CPR) as a proactive measure.
Oxidant Demand
Oxidants are the non-living contaminants in your water. The most common, by far, are non-living organics like sunscreen, cosmetics, lotions, deodorants, body oils, sweat, etc. Then there are the nitrogen compounds like nitrites, urea and ammonia, which eventually get reduced down into nitrates and other various disinfection byproducts. And depending on your tap water, metals can also be a major source of oxidant demand. Iron in particular can be a real nuisance.
How to minimize oxidant demand
Managing the oxidant demand dramatically reduces the overall chlorine demand.

AAD enzyme purge
Depending on the oxidant type, there are several things that can help. Just like sanitizer demand, supplementing chlorine with secondary oxidation really helps. This does not include UV, because UV is not an oxidizer. We are talking about Ozone and AOP systems here. These systems help destroy non-living organics, nitrogen compounds, and pretty much anything else that can be oxidized. As mentioned earlier, we strongly recommend supplementing chlorine with enzymes like Amino Acid Digester (AAD). AAD breaks down and removes oils, sunscreen, cosmetics, mucous and most of the other things that come off our bodies. And because enzymes circulate alongside chlorine throughout the entire system, their benefit is 24/7 in every corner of the pool.
As for metals like iron, copper and manganese, they should ideally be pre-filtered out from the tap water fill line. If that’s not an option and metals are already in your pool, you can bet high iron levels will consume chlorine rapidly, and leave behind brown-tinted water or ugly stains. If the metals are in your pool, you can either sequester them or chelate them with Metal and Scale Inhibitor (MSI). But be aware, the first time you use MSI, its purge dose can wipe out your chlorine levels entirely for a few days (talk about a temporary chlorine demand!). That’s because it takes time for a chelating agent like MSI to find all the metals and minerals (like calcium) to bind to. Once it does its job, MSI coexists with chlorine just fine.
Conclusion
The vast majority of chlorine demand is from non-living organics, aka bather waste. The second biggest contributor is nitrogen compounds like urea and ammonia, which require a lot of chlorine to oxidize out of the pool. Then metals. Then there’s the sanitizer demand, which, unless you’re facing an algae outbreak, is proportionally much less prevalent in the water than the oxidant demand. Most of the sanitizer demand is small (but critically important for health and safety!), and chlorine is made for killing germs.
To reduce chlorine demand, consider supplementing chlorine with secondary oxidizers/disinfection systems, and enzymes. For metals in particular, MSI is a good option for chelation to prevent oxidation.






 The second way to minimize sanitizer demand is to limit the growth and reproduction rate of algae. At any given time, an outdoor pool will be faced with algae spores trying to produce a colony. There is almost certainly more algae than there are germs, so we want to slow their growth and reproduction rates so chlorine can stay ahead. This can be accomplished by using
The second way to minimize sanitizer demand is to limit the growth and reproduction rate of algae. At any given time, an outdoor pool will be faced with algae spores trying to produce a colony. There is almost certainly more algae than there are germs, so we want to slow their growth and reproduction rates so chlorine can stay ahead. This can be accomplished by using 



 Source water is different everywhere you go, which is why we insist on testing tap water before filling a pool, or even servicing the pool. If you don’t know what is coming out of the tap, you will be at a severe disadvantage when it comes to managing pool chemistry. Since most pool operators test only a few factors–pH, total alkalinity, chlorine, and sometimes calcium hardness–otherwise predictable problems occur. To really grasp what will happen to the pool, one must know what is coming out of the tap.
Source water is different everywhere you go, which is why we insist on testing tap water before filling a pool, or even servicing the pool. If you don’t know what is coming out of the tap, you will be at a severe disadvantage when it comes to managing pool chemistry. Since most pool operators test only a few factors–pH, total alkalinity, chlorine, and sometimes calcium hardness–otherwise predictable problems occur. To really grasp what will happen to the pool, one must know what is coming out of the tap.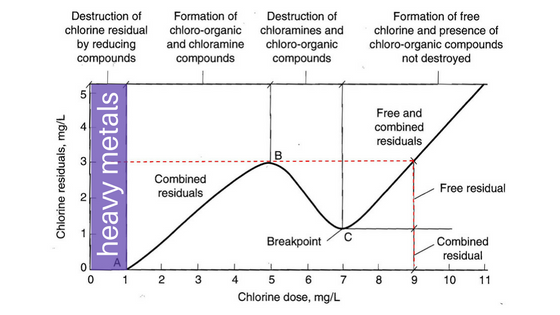
 Metal stains in pools can be prevented. The way to prevent metal stains is to control metals. There are a few ways to control metals, but for the purposes of this article, let’s stay with what is practical in the pool business: removal, sequestration, and chelation.
Metal stains in pools can be prevented. The way to prevent metal stains is to control metals. There are a few ways to control metals, but for the purposes of this article, let’s stay with what is practical in the pool business: removal, sequestration, and chelation.



 The first thing is to address bather waste with something other than chlorine. We recommend a commercial-grade enzyme like our
The first thing is to address bather waste with something other than chlorine. We recommend a commercial-grade enzyme like our